Summary
Background
The pandemic of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) might be curtailed by vaccination. We assessed the safety, reactogenicity, and immunogenicity of a viral vectored coronavirus vaccine that expresses the spike protein of SARS-CoV-2.
Methods
Findings
Between April 23 and May 21, 2020, 1077 participants were enrolled and assigned to receive either ChAdOx1 nCoV-19 (n=543) or MenACWY (n=534), ten of whom were enrolled in the non-randomised ChAdOx1 nCoV-19 prime-boost group. Local and systemic reactions were more common in the ChAdOx1 nCoV-19 group and many were reduced by use of prophylactic paracetamol, including pain, feeling feverish, chills, muscle ache, headache, and malaise (all p<0·05). There were no serious adverse events related to ChAdOx1 nCoV-19. In the ChAdOx1 nCoV-19 group, spike-specific T-cell responses peaked on day 14 (median 856 spot-forming cells per million peripheral blood mononuclear cells, IQR 493–1802; n=43). Anti-spike IgG responses rose by day 28 (median 157 ELISA units [EU], 96–317; n=127), and were boosted following a second dose (639 EU, 360–792; n=10). Neutralising antibody responses against SARS-CoV-2 were detected in 32 (91%) of 35 participants after a single dose when measured in MNA80 and in 35 (100%) participants when measured in PRNT50. After a booster dose, all participants had neutralising activity (nine of nine in MNA80 at day 42 and ten of ten in Marburg VN on day 56). Neutralising antibody responses correlated strongly with antibody levels measured by ELISA (R2=0·67 by Marburg VN; p<0·001).
Interpretation
ChAdOx1 nCoV-19 showed an acceptable safety profile, and homologous boosting increased antibody responses. These results, together with the induction of both humoral and cellular immune responses, support large-scale evaluation of this candidate vaccine in an ongoing phase 3 programme.
Funding
UK Research and Innovation, Coalition for Epidemic Preparedness Innovations, National Institute for Health Research (NIHR), NIHR Oxford Biomedical Research Centre, Thames Valley and South Midland’s NIHR Clinical Research Network, and the German Center for Infection Research (DZIF), Partner site Gießen-Marburg-Langen.
Evidence before this study
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was identified as the causative agent of COVID-19 in January, 2020. There are currently no licensed vaccines to prevent COVID-19. ChAdOx1 nCoV-19 has previously been reported to be immunogenic and protective against pneumonia in a rhesus macaque challenge model. We searched PubMed for research articles published between database inception and July 6, 2020, using the terms “SARS-CoV-2”, “vaccine”, “clinical trial”, and “phase”. No language restrictions were applied. We identified one published clinical trial, describing a trial done in China of an adenovirus-5-vectored vaccine against SARS-CoV-2, using a single dose at three different dose levels. The vaccine was tolerated, with reactogenicity increased at the highest dose. Antibodies, neutralising antibodies in a proportion of vaccinees, and cellular responses were induced. A further clinical trial, which was done in the USA, has been reported on medRxiv. The vaccine was a lipid nanoparticle-formulated, nucleoside-modified, mRNA vaccine that encodes trimerised SARS-CoV-2 spike glycoprotein receptor binding domain administered at one or two doses of three dose levels. The vaccine was tolerated, with reactogenicity increased at the highest dose. Antibodies and neutralising antibodies were induced in a dose-dependent manner and increased after a second dose.
Added value of this study
We report the results of the first clinical study of ChAdOx1 nCoV-19 (AZD1222). The vaccine was safe and tolerated, with reduced reactogenicity when paracetamol was used prophylactically for the first 24 h after vaccination. Reactogenicity was reduced after a second dose. Humoral responses to SARS-CoV-2 spike protein peaked by day 28 post prime and cellular responses were induced in all participants by day 14. Neutralising antibodies were induced in all participants after a second vaccine dose. After two doses, potent cellular and humoral immunogenicity was present in all participants studied.
Implications of all the available evidence
A vaccine against SARS-CoV-2 could be used to prevent infection, disease, and death in the global population, with high-risk populations such as hospital workers and older adults (eg, ≥65 years of age) prioritised to receive vaccination. The immune correlates of protection against SARS-CoV-2 have not yet been determined. Immunisation with ChAdOx1 nCoV-19 results in rapid induction of both humoral and cellular immune responses against SARS-CoV-2, with increased responses after a second dose. Further clinical studies, including in older adults, should be done with this vaccine.
Introduction
The pandemic has placed substantial pressures on health systems delivering care for patients with COVID-19 and caused disruption of non-COVID-19 health-care provision, in addition to negative effects on the global economy. Further health consequences are anticipated.
An ideal vaccine against SARS-CoV-2 would be effective after one or two vaccinations; would protect target populations such as older adults and those with comorbidities, including immunocompromised individuals; would confer protection for a minimum of 6 months; and would reduce onward transmission of the virus to contacts. Replication-deficient viral vectored vaccines have been used in immunocompromised individuals with no safety concerns
,
,
and ChAdOx1 vaccines are immunogenic in older adults
and can be manufactured at large scale, making this platform technology a promising candidate to develop a vaccine for the prevention of COVID-19. Adenoviral vectors have previously been combined with DNA and poxviral vectors to attempt to improve immunogenicity, with adenovirus or modified vaccinia virus Ankara prime-boost regimens showing enhancement of both cellular and humoral immunity. Use of homologous adenoviral regimens has largely been avoided because of presumed induction of antivector immunity, inhibiting the potency of a second dose.
and data from a phase 1 clinical trial showed that ChAdOx1 MERS was safe and well tolerated at all three doses tested (5 × 109 viral particles, 2·5 × 1010 viral particles, and 5 × 1010 viral particles).
In addition, the highest dose elicited both humoral and cellular responses against MERS-CoV in all vaccinees within 1 month of vaccination.
The ChAdOx1 nCoV-19 vaccine (AZD1222) consists of the replication-deficient simian adenovirus vector ChAdOx1, containing the full-length structural surface glycoprotein (spike protein) of SARS-CoV-2, with a tissue plasminogen activator leader sequence. ChAdOx1 nCoV-19 expresses a codon-optimised coding sequence for the spike protein (GenBank accession number MN908947).
We did a phase 1/2 single-blind, randomised controlled trial of ChAdOx1 nCoV-19 compared with a licensed meningococcal group A, C, W-135, and Y conjugate vaccine (MenACWY; Nimenrix, Pfizer, UK), as control vaccine, in healthy adults in the UK. In this preliminary report, we describe the immunogenicity, reactogenicity, and safety of vaccination with 5 × 1010 viral particles of ChAdOx1 nCoV-19 in single-dose and two-dose regimens.
Methods
Study design and participants
Written informed consent was obtained from all participants, and the trial is being done in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice. This study was approved in the UK by the Medicines and Healthcare products Regulatory Agency (reference 21584/0424/001-0001) and the South Central Berkshire Research Ethics Committee (reference 20/SC/0145). Vaccine use was authorised by Genetically Modified Organisms Safety Committees at each participating site.
Randomisation and masking
Participants were randomly assigned (1:1) to receive either the ChAdOx1 nCoV-19 vaccine or the MenACWY vaccine. MenACWY was used as a comparator vaccine to maintain blinding of participants who experienced local or systemic reactions, since these reactions are a known association with viral vector vaccinations. Use of saline as a placebo would risk unblinding participants as those who had notable reactions would know they were in the ChAdOx1 nCoV-19 vaccine group.
Randomisation lists, using block randomisation stratified by study group and study site, were generated by the study statistician (MV). Block sizes of two and four were chosen to align with the study group sizes and the sequence of enrolment, and varied across study groups. Computer randomisation was done with full allocation concealment within the secure web platform used for the study electronic case report form (REDCap version 9.5.22; Vanderbilt University, Nashville, TN, USA). The trial staff administering the vaccine prepared vaccines out of sight of the participants and syringes were covered with an opaque material until ready for administration to ensure blinding of participants. Clinical investigators and the laboratory team remained blinded to group allocation.
Procedures
The vaccine was manufactured according to current Good Manufacturing Practice by the Clinical BioManufacturing Facility (University of Oxford, Oxford, UK) as previously described,
with only minor modifications, as described in the Investigational Medicinal Product Dossier and approved by the regulatory agency in the UK. ChAdOx1 nCoV-19 was administered at a dose of 5 × 1010 viral particles. The MenACWY vaccine was provided by the UK Department of Health and Social Care and administered as per summary of product characteristics at the standard dose of 0·5 mL. Vaccines were administered as a single intramuscular injection into the deltoid.
Participants in all groups had blood samples drawn and clinical assessments for safety as well as immunology at days 0 and 28, and will also be followed up at days 184 and 364. A later amendment to the protocol (amendment date June 22, 2020) provided for additional testing of booster vaccinations in a subset of participants, the results of which are not yet available and are not included in this Article.
In two of the five trial sites (Oxford and Southampton), a protocol amendment (amendment date May 6, 2020) was implemented to allow prophylactic paracetamol to be administered before vaccination and participants were advised to continue with 1 g of paracetamol every 6 h for 24 h to reduce vaccine-associated reactions. All participants enrolled after the protocol amendment at these two sites were given prophylactic paracetamol and randomised equally to the vaccine or control arms of the study.
Participants were observed in the clinic for 30–60 min after the vaccination procedure and were asked to record any adverse events using electronic diaries during the 28-day follow-up period. Expected and protocol-defined local site reactions (injection site pain, tenderness, warmth, redness, swelling, induration, and itch) and systemic symptoms (malaise, muscle ache, joint pain, fatigue, nausea, headache, chills, feverishness [ie, a self-reported feeling of having a fever], and objective fever defined as an oral temperature of 38°C or higher) were recorded for 7 days. All other events were recorded for 28 days and serious adverse events are recorded throughout the follow-up period.
Severity of adverse events are graded with the following criteria: mild (transient or mild discomfort for <48 h, no interference with activity, and no medical intervention or therapy required), moderate (mild to moderate limitation in activity [some assistance might be needed] and no or minimal medical intervention or therapy required), severe (marked limitation in activity [some assistance usually required] and medical intervention or therapy required), and potentially life-threatening (requires assessment in emergency department or hospitalisation). Unsolicited adverse events are reviewed for causality by two clinicians blinded to group allocation, and events considered to be possibly, probably, or definitely related to the study vaccines were reported. Laboratory adverse events were graded by use of site-specific toxicity tables, which were adapted from the US Food and Drug Administration toxicity grading scale.
Convalescent plasma samples from adults (≥18 years) with PCR-positive SARS-CoV-2 infection were obtained from symptomatic patients admitted to hospital or from surveillance on health-care workers who did not have symptomatic infection. These samples were tested using standardised ELISA, MIA, PseudoNA, and Marburg VN. Different samples were analysed across the assays, dependent on sample availability, laboratory capacity, and assay-specific requirements. Where multiple longitudinal samples were available for the same participant, only one timepoint is included in the analyses in this Article and the earliest timepoint (at least 20 days after initial symptoms) was selected.
Outcomes
The convalescent sample collection of PCR-positive hospitalised patients with COVID-19 or asymptomatic health-care workers was done to characterise the immunological properties of COVID-19 and not for the purposes of the clinical trial (Gastrointestinal Illness in Oxford: COVID substudy [Sheffield Research Ethics Committee reference: 16/YH/0247], ISARIC/WHO Clinical Characterisation Protocol for Severe Emerging Infections [Oxford Research Ethics Committee C reference 13/SC/0149], and Sepsis Immunomics project [Oxford Research Ethics Committee C, reference 19/SC/0296]).
Statistical analysis
Safety endpoints are described as frequencies (%) with 95% binomial exact CIs. Medians and IQRs are presented for immunological endpoints and analyses are considered descriptive only, as the full set of samples have not yet been analysed on all platforms and therefore results reported here are preliminary. Participants were analysed according to the group to which they were randomised. To assess the effect of prophylactic paracetamol use, the occurence of adverse reactions in the first 2 days after vaccination was analysed as a binary variable using adjusted logistic regression with results presented as adjusted odds ratios. The model adjusted for age, sex, occupation (health-care worker or not), smoking, alcohol consumption, and body-mass index. To assess the relationship between responses on different assays, linear regression was used to analyse log-transformed post-baseline values. Statistical analyses were done using SAS version 9.4 and R version 3.6.1 or later.
Role of the funding source
The funders of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results

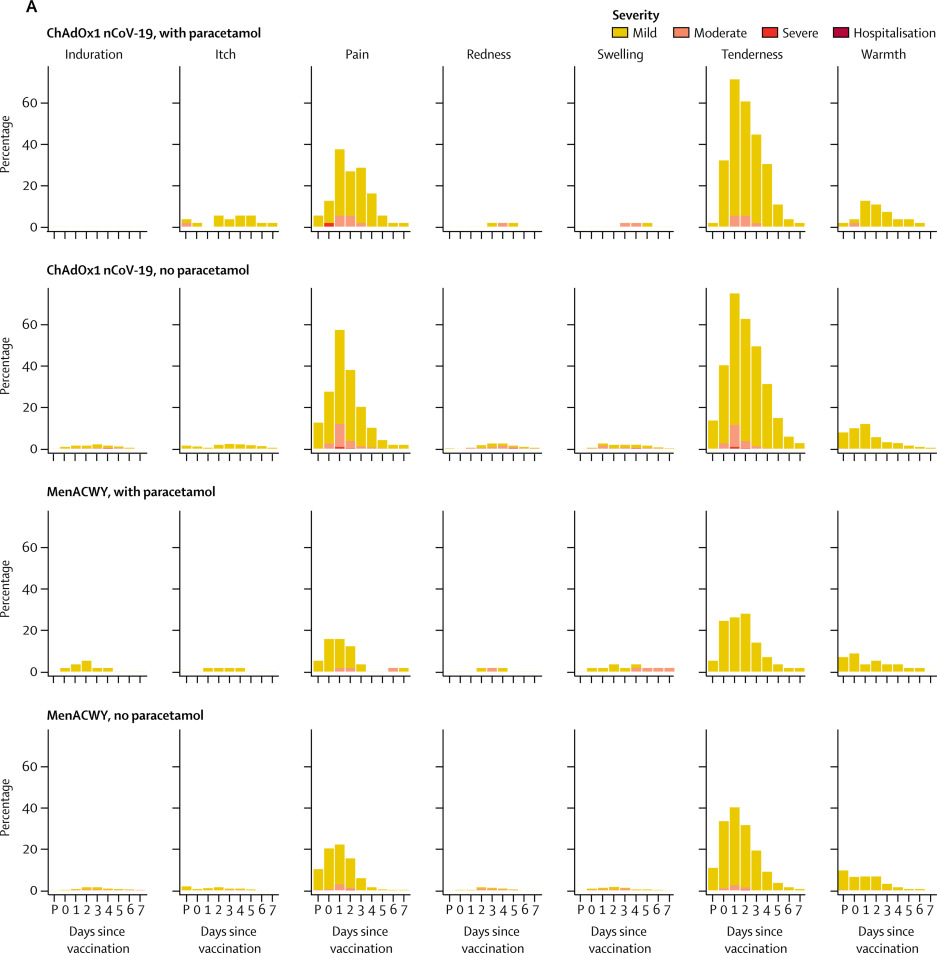
Figure 1Solicited local (A) and systemic (B) adverse reactions in first 7 days after vaccination as recorded in participant symptom electronic diaries
Day 0 is the day of vaccination. P=60-min post-vaccination observation period in the clinic. MenACWY=meningococcal group A, C, W-135, and Y conjugate vaccine. *Mild: 38·0°C to <38·5°C; moderate: 38·5°C to <39·0°C; severe: ≥39·0°C. †Self-reported feeling of feverishness.

Figure 1Solicited local (A) and systemic (B) adverse reactions in first 7 days after vaccination as recorded in participant symptom electronic diaries
Day 0 is the day of vaccination. P=60-min post-vaccination observation period in the clinic. MenACWY=meningococcal group A, C, W-135, and Y conjugate vaccine. *Mild: 38·0°C to <38·5°C; moderate: 38·5°C to <39·0°C; severe: ≥39·0°C. †Self-reported feeling of feverishness.
Fatigue and headache were the most commonly reported systemic reactions. Fatigue was reported in the ChAdOx1 nCoV-19 group by 340 (70%) participants without paracetamol and 40 (71%) with paracetamol and in the MenACWY group by 227 (48%) participants without paracetamol and 26 (46%) with paracetamol, whereas headaches were reported in the ChAdOx1 nCoV-19 group by 331 (68%) participants without paracetamol and 34 (61%) with paracetamol and in the MenACWY group by 195 (41%) participants without paracetamol and 21 (37%) participants with paracetamol.

Figure 2Solicited local (A) and systemic (B) adverse reactions in first 7 days after priming and booster doses of ChAdOx1 nCoV-19 in the non-randomised subset of ten participants
Day 0 is the day of vaccination. P=60-min post-vaccination observation period in the clinic. *Mild: 38·0°C to <38·5°C; moderate: 38·5°C to <39·0°C; severe: ≥39·0°C. †Self-reported feeling of feverishness.

Figure 3SARS-CoV-2 IgG response by standardised ELISA to spike protein in trial participants (A) and in 180 convalescent plasma samples from 172 patients with PCR-confirmed COVID-19 and eight asymptomatic health-care workers (B)

Figure 4Live SARS-CoV-2 neutralisation assays (Marburg VN and PHE PRNT50) and microneutralisation assays (PHE MNA)

Figure 5PseudoNA results in trial participants and in convalescent plasma samples from 146 patients with PCR-confirmed COVID-19 and 24 asymptomatic health-care workers
Solid lines connect samples from the same participant. Boxes show median (IQR). Results for days 35 and 42 are samples from participants who received a booster dose at day 28. IC=inhibitory concentration. MenACWY=meningococcal group A, C, W-135, and Y conjugate vaccine.

Figure 6Interferon-γ ELISpot response to peptides spanning the SARS-CoV-2 spike vaccine insert
Error bars show median (IQR). The lower limit of detection, indicated with the dotted line, is 48 spot-forming cells per million PBMCs. PBMC=peripheral blood mononuclear cell. SARS-CoV-2=severe acute respiratory syndrome coronavirus 2. ELISpot=enzyme linked immunospot. MenACWY=meningococcal group A, C, W-135, and Y conjugate vaccine.
A small number (four [4%] of 98) participants had neutralising antibody titres greater than 8 against SARS-CoV-2 spike protein before vaccination (Marburg VN) and 11 (4%) of 270 participants had high ELISA titres at baseline, representing possible prior asymptomatic infection.
Discussion
,
,
,
at this dose level, as well as to some licensed vaccines.
A dose of 5 × 1010 viral particles was chosen on the basis of our previous experience with ChAdOx1 MERS, where despite increased reactogenicity, a dose–response relationship with neutralising antibodies was observed.
The protocol was written when the pandemic was accelerating in the UK and a single higher dose was chosen to provide the highest chance of rapid induction of neutralising antibody. In the context of a pandemic wave where a single higher, but more reactogenic, dose might be more likely to rapidly induce protective immunity, the use of prophylactic paracetamol appears to increase tolerability and would reduce confusion with COVID-19 symptoms that might be caused by short-lived vaccine-related symptoms without compromising immunogenicity.
We show that a single dose of ChAdOx1 nCoV-19 elicits an increase in spike-specific antibodies by day 28 and neutralising antibody in all participants after a booster dose. High levels of neutralising antibody at baseline seen in a small number of participants probably indicates prior asymptomatic infection, as potential participants with recent COVID-19-like symptoms or with a history of positive PCR test for SARS-CoV-2 were excluded from the study. Individuals with high titres on the day of vaccination who received ChAdOx1 nCoV-19 were boosted by vaccination.
Although a correlate of protection has not been defined for COVID-19, high levels of neutralising antibodies have been shown in convalescent individuals, with a wide range, as confirmed in our study.
,
Neutralising antibody titres were increased by a two-dose regimen, and further investigation of this approach is underway. The correlation of neutralisation assays with IgG quantitation indicates that, if confirmed, a standardised ELISA might be sufficient to predict protection, should neutralising antibody also be shown to be protective in humans. We have presented data from three different live neutralising antibody assays and a pseudo-neutralisation assay, which show tight correlation with each other but give very different neutralising antibody titres. This issue highlights the urgent need for centralised laboratory infrastructure to allow bridging between vaccine candidates and accelerate the availability of multiple products to provide the global capacity to end the pandemic. If any one candidate demonstrates efficacy, bridging this result to other candidate vaccines through rigorously conducted laboratory assays will become a crucial issue for global health.
,
,
Adenovirus-vectored vaccines are known to induce strong cellular immunity and ChAdOx1 nCoV-19 vaccination resulted in marked increases in SARS-CoV-2 spike-specific effector T-cell responses as early as day 7, peaking at day 14 and maintained up to day 56 as expected with adenoviral vectors. However, a boost in cellular responses was not observed following the second ChAdOx1 nCoV-19 dose. This is consistent with previous findings on viral vectored vaccines given as part of a homologous prime-boost regimen.
As previously reported,
anti-vector immunity was low before vaccination in UK adults aged 18–55 years, with no relationship between the presence of antibodies to ChAdOx1 and immune response to the vaccine antigen. Future studies will address the potential effect of anti-vector antibodies on homologous boosting, although in the subgroup reported on here, who received two vaccinations 28 days apart, there was clear evidence of boosting of antibody response to SARS-CoV-2 spike protein.
,
,
In conclusion, ChAdOx1 nCoV-19 was safe, tolerated, and immunogenic, while reactogenicity was reduced with paracetamol. A single dose elicited both humoral and cellular responses against SARS-CoV-2, with a booster immunisation augmenting neutralising antibody titres. The preliminary results of this first-in-human clinical trial supported clinical development progression into ongoing phase 2 and 3 trials. Older age groups with comorbidities, health-care workers, and those with higher risk for SARS-CoV-2 exposure are being recruited and assessed for efficacy, safety, and immunogenicity of ChAdOx1 nCoV-19 given as a single-dose or two-dose administration regimen in further trials conducted in the UK and overseas. We will also evaluate the vaccine in children, once sufficient safety data have been accumulated in adult studies. Phase 3 trials are now underway in Brazil, South Africa, and the UK and will evaluate vaccine efficacy in diverse populations.
Contributors
SCG and AJP conceived and designed the trial and AJP is the chief investigator. AJP, PMF, DJ, HR, and MV contributed to the protocol and design of the study. AF, PH, RL, KMP, SNF, BA, and AVSH were the study site principal investigators. DB, MB, CD, SBi, SBe, EAC, TL, KJE, ALF, BH, RM, and SB-R were responsible for laboratory testing and assay development. PKA, DJ, HR, PMF, AMM, MR, and MS contributed to the implementation of the study. MV conducted the statistical analysis. CG, ADD, and RT were responsible for vaccine manufacturing. TL and SCG were responsible for vaccine development. AVSH and SCG developed the ChAdOx1 vector. TL, KJE, MV, SCG, AVSH, PMF, and AJP contributed to the preparation of the report. All other authors contributed to the implementation of the study and data collection. All authors critically reviewed and approved the final version.
Declaration of interests
SCG is co-founder and board member of Vaccitech (collaborators in the early development of this vaccine candidate) and named as an inventor on a patent covering use of ChAdOx1-vectored vaccines and a patent application covering this SARS-CoV-2 vaccine. TL is named as an inventor on a patent application covering this SARS-CoV-2 vaccine and consultant to Vaccitech. PMF is a consultant to Vaccitech. AJP is Chair of the UK Department of Health and Social Care’s Joint Committee on Vaccination & Immunisation (JCVI), but does not participate in policy advice on coronavirus vaccines, and is a member of the WHO Strategic Advisory Group of Experts (SAGE). AVSH is a co-founder of and consultant to Vaccitech and is named as an inventor on a patent covering design and use of ChAdOx1-vectored vaccines. AF is a member of JCVI, Chair of the WHO European Technical Advisory Group of Experts on Immunisation, an ex-officio member of WHO SAGE working group on COVID-19 vaccines, and acting director of National Institute for Health Research West of England Local Clinical Research Network. KMP reports grants from the NIHR Imperial Biomedical Research Centre and Gilead Sciences, and personal fees from Sanofi Pasteur, outside of the submitted work. MS reports grants from Janssen, GlaxoSmithKline, Medimmune, Novavax, and MCM and grants and non-financial support from Pfizer, outside of the submitted work. CG reports personal fees from the Duke Human Vaccine Institute, outside of the submitted work. ADD reports grants and personal fees from AstraZeneca, outside of the submitted work. In addition, ADD has a patent manufacturing process for ChAdOx vectors with royalties paid to AstraZeneca, and a patent ChAdOx2 vector with royalties paid to AstraZeneca. The other authors declare no competing interests.
Data sharing
Acknowledgments
Supplementary Material
References
- 1.
Coronavirus disease (COVID-19) situation report—181.
World Health Organization,
Geneva2020 - 2.
Draft Landscape of COVID-19 candidate vaccines.
- 3.
Antiretroviral therapy alone versus antiretroviral therapy with a kick and kill approach, on measures of the HIV reservoir in participants with recent HIV infection (the RIVER trial): a phase 2, randomised trial.
Lancet. 2020; 395: 888-898
- 4.
Aiming for protective T-cell responses: a focus on the first generation conserved-region HIVconsv vaccines in preventive and therapeutic clinical trials.
Expert Rev Vaccines. 2019; 18: 1029-1041
- 5.
Therapeutic vaccination refocuses T-cell responses towards conserved regions of HIV-1 in early treated individuals (BCN 01 study).
EClinicalMedicine. 2019; 11: 65-80
- 6.
Heterologous two-dose vaccination with simian adenovirus and poxvirus vectors elicits long-lasting cellular immunity to influenza virus A in healthy adults.
EBioMedicine. 2018; 29: 146-154
- 7.
A single dose of ChAdOx1 MERS provides protective immunity in rhesus macaques.
Sci Adv. 2020; 6eaba8399
- 8.
Safety and immunogenicity of a candidate Middle East respiratory syndrome coronavirus viral-vectored vaccine: a dose-escalation, open-label, non-randomised, uncontrolled, phase 1 trial.
Lancet Infect Dis. 2020; 20: 816-826
- 9.
ChAdOx1 nCoV-19 vaccination prevents SARS-CoV-2 pneumonia in rhesus macaques.
BioRxiv. 2020; ()
- 10.
A novel chimpanzee adenovirus vector with low human seroprevalence: improved systems for vector derivation and comparative immunogenicity.
PLoS One. 2012; 7e40385
- 11.
Laboratory-scale production of replication-deficient adenovirus vectored vaccines.
Methods Mol Biol. 2016; 1349: 121-135
- 12.
Assessment of novel vaccination regimens using viral vectored liver stage malaria vaccines encoding ME-TRAP.
Sci Rep. 2018; 83390
- 13.
Safety and immunogenicity of a novel recombinant simian adenovirus ChAdOx2 as a vectored vaccine.
Vaccines (Basel). 2019; 7: 40
- 14.
A monovalent chimpanzee adenovirus ebola vaccine boosted with MVA.
N Engl J Med. 2016; 374: 1635-1646
- 15.
Postlicensure safety surveillance of recombinant zoster vaccine (Shingrix)—United States, October 2017–June 2018.
MMWR Morb Mortal Wkly Rep. 2019; 68: 91-94
- 16.
DNA vaccine protection against SARS-CoV-2 in rhesus macaques.
Science. 2020; ()
- 17.
Convergent antibody responses to SARS-CoV-2 in convalescent individuals.
Nature. 2020; ()
- 18.
Neutralizing antibodies responses to SARS-CoV-2 in COVID-19 inpatients and convalescent patients.
Clin Infect Dis. 2020; ()
- 19.
Primary exposure to SARS-CoV-2 protects against reinfection in rhesus macaques.
Science. 2020; ()
- 20.
Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals.
Cell. 2020; 181 (): 1489
- 21.
Robust T cell immunity in convalescent individuals with asymptomatic or mild COVID-19.
bioRxiv. 2020; ()
- 22.
Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome.
Sci Immunol. 2020; 5eabd2071
- 23.
A double-inactivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge.
J Virol. 2011; 85: 12201-12215
- 24.
Anti-spike IgG causes severe acute lung injury by skewing macrophage responses during acute SARS-CoV infection.
JCI Insight. 2019; 4123158
- 25.
Immunization with modified vaccinia virus Ankara-based recombinant vaccine against severe acute respiratory syndrome is associated with enhanced hepatitis in ferrets.
J Virol. 2004; 78: 12672-12676
Article Info
Publication History
Identification
Copyright
© 2020 The Author(s). Published by Elsevier Ltd.
User License
Creative Commons Attribution – NonCommercial – NoDerivs (CC BY-NC-ND 4.0) |

Permitted
For non-commercial purposes:
- Read, print & download
- Redistribute or republish the final article
- Text & data mine
- Translate the article (private use only, not for distribution)
- Reuse portions or extracts from the article in other works
Not Permitted
- Sell or re-use for commercial purposes
- Distribute translations or adaptations of the article