1. Introduction
], with elevated levels of hepatitis C (HCV) co-infection occurring among HIV-positive MSM in high-income countries [
], but much lower transmission occurring among HIV-negative MSM [
,
. This recent MSM HCV epidemic has been associated with sexual and drug-related behaviours [
], with the polarised pattern of HCV in HIV-infected MSM likely due to heterogeneity in risk behaviours and HIV sero-adaptive behaviours [
]. Over 2012–2017, HIV incidence has halved in the UK [
] mainly due to the UK achieving very high levels of antiretroviral therapy (ART) coverage and HIV viral suppression [
].
,
. Many countries are expanding the availability of PrEP among MSM [
]. However, as occurred with the expansion of ART [
], there are concerns that PrEP use could result in increased sexual risk taking, thus increasing the transmission of sexually transmitted infections (STIs), including HCV [
]. Recent studies have confirmed this, giving a consistent picture that the incidence of STIs increases following initiation of PrEP [
,
.
]. Elimination initiatives are attempting to achieve this goal among MSM, mainly targeting HIV-diagnosed MSM [
–
,
]. In the UK, HIV-diagnosed MSM are advised to be screened for HCV each year [], while HIV-negative MSM are rarely tested [] and there are no HCV testing guidelines for MSM using PrEP.
,
], but none have accounted for the HCV transmission dynamics among HIV-negative MSM. In this study, we use modelling to determine what HCV testing and treatment strategies are needed to reduce the overall incidence of HCV among MSM by 90% by 2030 in the UK. We assess how PrEP and any associated changes in condom use may affect the impact achieved, and determine the need for HCV screening among PrEP users and other HIV-negative MSM to help inform future policy and guidelines.
2. Methods
2.1 Model derivation
] to include PrEP use (details in Supplementary material). The model (Supplementary Fig. S1) stratifies MSM by: HIV and PrEP status (susceptible on/off PrEP, acute HIV infection on/off PrEP, undiagnosed or diagnosed chronic HIV infection); HCV-status (susceptible, acute HCV infection, undiagnosed and diagnosed chronic HCV infection); and either low- or high-risk sexual behaviour, defined by the annual number of anal sex partners (high-risk defined as ≥ 15). Individuals are not assumed to change their risk.
]. MSM also mix preferentially, more commonly choosing partners of the same sexual risk and HIV-status (see Supplementary materials).
,
,
. PrEP was assumed to scale-up from 2018, with the coverage of PrEP reaching 12·5% of HIV-negative MSM by 2020 with the average duration on PrEP being 8·2 months [
]. PrEP users are screened quarterly for HIV and upon a positive diagnosis stop using PrEP. This frequent HIV-testing means PrEP users are diagnosed before reaching chronic HIV infection [
]. Non-PrEP users who acquire acute HIV infection, firstly transition to undiagnosed HIV infection and are then diagnosed at current UK HIV-testing rates (2·3 years between infection and diagnosis) [
]. Acute HIV is assumed to have elevated HIV transmission risk (26-fold) [
]. A proportion of HIV-diagnosed MSM are on ART, which increases survival 3·4-fold, with a proportion being virally supressed and having negligible HIV transmission risk (see Supplementary materials).
]. At baseline, we assume 2.2 years from diagnosis to completing HCV treatment, consistent with UK data for pre-DAA treatments [
]. Before 2015, we assume different cure rates for HIV-positive (sustained viral response or SVR of 35–42%) [
] and HIV-negative MSM (SVR of 59–69%) [
] based on pre-DAA treatments, but then assume higher cure rates from 2015 for DAA therapies (SVR of 90–100%) [
]. From 2018, we then consider the impact of various scenarios of improved HCV screening with faster linkage-to-treatment following diagnosis (6 months to treatment completion); more consistent with current treatment rates [
]. This assumes a 3-month waiting time and an HCV treatment duration of 8–12 weeks. MSM failing treatment are retreated at the same rate as initial HCV treatment.
2.2 Parameterization of sexual risk behaviour
,
] EMIS was a pan-European internet survey on interventions, needs, behaviours and morbidities regarding HIV and STI transmission among MSM. Individuals could complete the survey online during Summer 2010. Over 180,000 men took part from 38 countries, including 18,000 in the UK [
]. From EMIS, we calculated the level of preferential mixing by HIV-status; proportion of MSM in the low and high-risk groups; prevalence of chem-sex; and levels of condom use stratified by the assumed HIV sero-concordancy. These model parameters are detailed in Supplementary Table S3, and summarised in Table 1. Briefly, EMIS data suggests 17·4% of MSM are high-risk, amongst whom the prevalence of chem-sex in last year is higher than among low-risk MSM (22.6% versus 11.5%). The baseline model assumed that MSM have sex more often with others of the same perceived HIV-status, with perceived HIV-positive concordant partnerships having lower condom use (13%) than other partnerships (68%) [
].
Table 1Key model parameters with ranges and details of estimation included. (For the full version see Supplementary Table S3.)
2.3 Baseline model calibration
]. UK CHIC involved a collaboration of UK centres providing care for people living with HIV (PLHIV). The study gathered data relating to clinical care of PLHIV since 1996, including data on HCV incidence and prevalence among HIV-diagnosed MSM.
] and a chronic HCV prevalence within the range 9·6–10·2% for HIV-infected MSM at equilibrium [
]. Model runs were only accepted if they also projected a prevalence of HCV among HIV-negative MSM within the range of 0·6–2·1% [
]. In total, we performed 668 runs to obtain 500 fits (75% acceptance rate). For our results, we present the 2·5% to 97·5% percentile range from these 500 model fits, denoted as the 95% central range (95% CR).
] to 98% [
]; (2) proportion of those on ART that are virally suppressed from 72% to 97%; and (3) HIV testing frequency from every 3·2 years [
] to 2·3 years [
]. The resulting model projections (Supplementary Figs. S7–S12) were then validated against data suggesting a 55·5% (95% CI 34·4–72·7%) decrease in the annual rate of new HIV infections among MSM over 2012–2017 in the UK [
]. HCV prevalence and incidence also decrease over this period due to an increase to DAA SVR rates from 2015 [
,
. Despite being not fit to this data, our model projections for HCV incidence among HIV-diagnosed MSM and HIV-negative MSM off PrEP for 2012 are comparable to UK data estimates from that period, as shown in Supplementary Figs. S7–S12 [
].
2.4 PrEP intervention scenarios
]. This coverage assumption was based on NHS-eligibility criteria, where only MSM recently participating in unprotected sex are eligible for PrEP. The relative coverage of PrEP among low- and high-risk MSM reflects this eligibility criteria (see Supplementary materials). The efficacy of PrEP for reducing the risk of HIV acquisition was assumed to be 91.5% (86–97%) [
,
,
.
,
,
,
, we only considered reductions in condom use among MSM using PrEP. In our modelling, we either assume no risk compensation (Scenario S0) or that all PrEP users halve their consistency of condom use from 68% to 34% with all partners (Scenario S1). This assumption is varied in our sensitivity analyses.
2.5 Model analyses
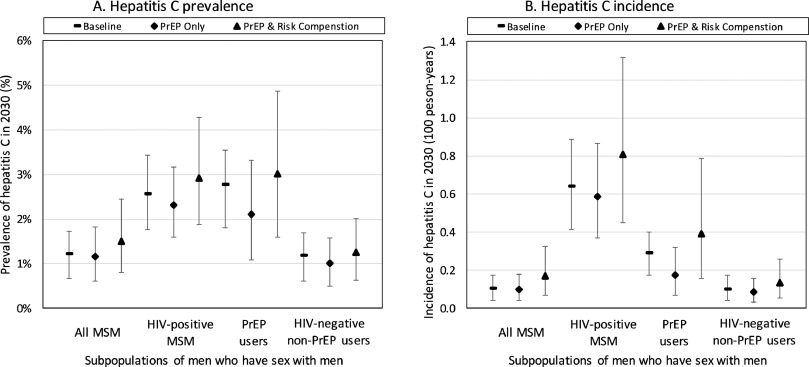
The main aim of the analysis is to determine what level of HCV screening and treatment is needed among different MSM sub-populations to eliminate HCV in all MSM, while incorporating the possible effects of PrEP scale-up. However, we firstly considered the possible impact of using DAAs as the new standard of care from 2015, and the effect that PrEP alone could have on the number of new HIV and HCV infections, as well as HCV prevalence and incidence in 2030 compared to 2018 levels.
We then evaluated the impact of different HCV screening and treatment scenarios initiated from 2020, to see what is needed to achieve the WHO elimination target. We firstly evaluated the impact of more frequent HCV screening for HIV-diagnosed MSM or PrEP users, with PrEP users otherwise having the same low level of HCV screening as HIV-negative MSM. The impact on HCV incidence among MSM using PrEP was estimated, with the relative change being compared to what the incidence was in those MSM in 2018. Similarly, the impact on HCV incidence in other groups was estimated. For these scenarios, we also assumed improved linkage-to-treatment for those MSM sub-groups with enhanced screening, with MSM completing treatment within 6 months of diagnosis. Lastly, we considered whether improved screening and linkage-to-treatment for HIV-negative MSM not using PrEP was needed to reach the elimination targets. For MSM using PrEP and HIV-diagnosed MSM, 3, 6 or 12-monthly screening were considered, while the screening frequency for HIV-negative MSM not using PrEP was fitted to give an overall 90% reduction in HCV incidence by 2030.
2.6 Uncertainty analysis
To ascertain which parameters are important for determining variability in the impact projections across the 500 baseline model fits, a linear regression analysis of covariance (ANCOVA) was performed on the projected decrease in overall HCV incidence (2018–2030) of undertaking 6-monthly screening among HIV-diagnosed MSM and MSM using PrEP (no risk compensation). The proportion of the sum of squares contributed by each parameter was calculated to determine each parameters’ importance to the variability in our projections.
We also performed a series of sensitivity analyses where we varied the following: (1) 4 versus 6 months between HCV diagnosis and treatment completion; (2) 25% versus 12·5% coverage of PrEP; (3) condom use among PrEP users decreases to 13% instead of 34% with risk compensation; (4) PrEP is distributed evenly between high and low-risk MSM, or to (5) just low-risk MSM or (6) just high-risk MSM; (7) no increased infectiousness of HCV with HIV co-infection; (8) 50% less mixing by HIV-status following PrEP introduction; (9) no increased risk associated with chem-sex; (10) HCV transmission rate decreased by 20% from 2012 to simulate a decreasing HCV epidemic before the introduction of DAA treatments, or conversely (11) increased by 20% to simulate an increasing HCV epidemic.
4. Discussion
]. Indeed, with 12·5% coverage of PrEP among HIV-negative MSM, yearly HCV screening of MSM on PrEP with rapid linkage-to-treatment could reduce the overall HCV incidence among MSM by up to 67·3% (95%CR 52·7–79·2%) over 2018 to 2030, with this increasing to 79·6% (95% CR 64·6–91·0%) if HIV-diagnosed MSM also receive equivalent screening and treatment. However, although considerable impact is possible from just reaching these groups, at this lower PrEP coverage (12.5%) the WHO HCV elimination targets cannot be reached without also improving screening (and linkage-to-treatment) among HIV-negative MSM not on PrEP to every 3–8 years. This changes, though, at higher PrEP coverage (25·0%), where only HIV-diagnosed MSM and PrEP users will require improved screening to reach the elimination target. Importantly, the required HCV screening frequencies in all MSM groups for achieving elimination are less than their average frequency of HIV testing in the UK [
,
, suggesting that these increases in HCV testing should be feasible if done at the same time as existing HIV testing using the same blood samples.
Our projections also highlight the importance of maintaining condom use among PrEP users, possibly through including counselling on STI and HCV risk for PrEP users as part of routine healthcare visits. Otherwise, reductions in condom use among PrEP users will increase HCV and STI transmission, especially among PrEP users and HIV-diagnosed MSM. Importantly, though, enhanced HCV screening in PrEP users may offset these additional HCV risks with our modelling suggesting similar impact can be achieved from our screening activities irrespective of the level of risk compensation.
The strength of our analysis is in modelling the full epidemic of HIV and HCV among MSM in the UK, while accounting for heterogeneities in sexual risk, patterns of mixing by HIV-status, and using detailed UK data. Despite these strengths, there are limitations.
], while it was relatively stable in the UK in 2012 [
]. Our sensitivity analyses suggest that it will be harder to control an increasing HCV epidemic, with a greater frequency of HCV screening being needed among HIV-negative MSM not on PrEP to reach the elimination targets than for a stable or decreasing HCV epidemic. Unfortunately, more precise parameterisation to specific settings is needed to form more detailed conclusions about the impact of our interventions’ in other settings. Additionally, our projections should not be generalised to lower and middle-income country settings which have different patterns of HIV and HCV prevalence among MSM [
].
,
; rather we assumed a stable HCV epidemic until 2012 (before the introduction of DAA treatments) as approximated by data from around that time. We also did not explicitly model the role of chem-sex; which we simplified by assuming a factor increase in transmission risk, reflective of what is found in the literature (see Supplementary materials). MSM were also assumed to have similar risk behaviour over their entire lifetime [
] due to insufficient data to parameterise transitions between risk groups over time.
]. However, because EMIS was widely distributed and undertaken by 18,000 men in UK, these biases should be less than for much smaller surveys undertaken in gay venues [
]. Our uncertainty analyses show the model is most sensitive to the increased infectiousness of HCV among HIV co-infected MSM and the level of heterogeneity in risk in the two risk groups. Better data on these parameters would improve our model projections. Importantly, we also cannot be certain of the exact scale-up of PrEP, nor the level of risk compensation that may occur among PrEP users. Our model projections suggest that uncertainty in the coverage of PrEP could have a large effect on our findings, while encouragingly, uncertainty in levels of risk compensation has little effect on the impact of large increases in screening.
,
,
,
. Our analysis builds on these previous studies by evaluating the level of HCV screening and treatment needed in all MSM sub-groups. Our analysis is novel in considering the effect of PrEP on HCV elimination targets, and the additional screening opportunities this provides. Our work compliments recent modelling from the US showing the beneficial impact that PrEP scale-up could have on STI transmission through more frequent STI screening at routine PrEP check-ups [
].
In the era of PrEP scale-up, our modelling suggests that at low PrEP coverage, 12, 6 or 3-monthly HCV screening of HIV-diagnosed MSM and PrEP users, alongside less frequent screening (every 3–8 years) of HIV-negative non-PrEP users, could achieve the WHO elimination target for decreasing HCV incidence by 2030 in the UK. At higher PrEP coverage, this may be achievable with just increased screening in HIV-diagnosed MSM and PrEP users. Importantly, these elimination targets are not possible through only screening HIV-diagnosed MSM (as most elimination initiatives are doing), with the scale-up of PrEP providing a valuable opportunity for increasing HCV-screening among higher-risk MSM. Importantly, though, this added impact of screening PrEP users relies on the assumption that PrEP users are in regular contact with care, which may not be the case if MSM acquire PrEP through unofficial channels. This emphasises the importance of making PrEP freely available through formal channels to ensure that HCV and other STIs can be tested and treated effectively.
], our findings highlight the need to look beyond just screening HIV-diagnosed MSM, to also screening PrEP users and HIV-negative non-PrEP users.
Partial presentation of results in meetings
N/A.
Financial support
Engineering and Physical Sciences Research Council (EPSRC) studentship for the Ph.D. studies of Louis MacGregor. This work was also partly funded by the Health Protection Research Unit in Evaluation of Interventions.
Acknowledgements
EMIS was funded by a grant of the European Commission under the EU Health Programme 2008-2013, as stipulated in Grant Agreement 2008 12 14 of the Executive Agency for Health and Consumers (EAHC). Further funding was received from CEEISCat (Centre d’Estudis Epidemiològics sobre les ITS/HIV/SIDA de Catalunya, Spain); Department of Health for England; Maastricht University (The Netherlands); Regione del Veneto (Italy); and Robert Koch Institute (Germany). Further funding was provided by: German Ministry of Health for the participation of men living in Ukraine and Moldova; Finnish Ministry of Health for Finland; Norwegian Institute of Public Health for Norway; Swedish Board of Health and Welfare for Sweden; and Bundeszentrale für gesundheitliche Aufklärung (BzGA) for German resident men.
MH and PV would also like to acknowledge support from the NIHR funded Health Protection Research Unit in Evaluation of Interventions. PV would also like to acknowledge the NIHR funded Health Protection Research Unit in STIs and BBVs. NM was supported by the National Institute for Drug Abuse [grant number R01 DA037773 ] and the University of California San Diego Center for AIDS Research (CFAR), a National Institute of Health (NIH) funded program [grant number P30 AI036214 ].