Summary
Background
Angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have been postulated to affect susceptibility to COVID-19. Observational studies so far have lacked rigorous ascertainment adjustment and international generalisability. We aimed to determine whether use of ACEIs or ARBs is associated with an increased susceptibility to COVID-19 in patients with hypertension.
Methods
In this international, open science, cohort analysis, we used electronic health records from Spain (Information Systems for Research in Primary Care [SIDIAP]) and the USA (Columbia University Irving Medical Center data warehouse [CUIMC] and Department of Veterans Affairs Observational Medical Outcomes Partnership [VA-OMOP]) to identify patients aged 18 years or older with at least one prescription for ACEIs and ARBs (target cohort) or calcium channel blockers (CCBs) and thiazide or thiazide-like diuretics (THZs; comparator cohort) between Nov 1, 2019, and Jan 31, 2020. Users were defined separately as receiving either monotherapy with these four drug classes, or monotherapy or combination therapy (combination use) with other antihypertensive medications. We assessed four outcomes: COVID-19 diagnosis; hospital admission with COVID-19; hospital admission with pneumonia; and hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis. We built large-scale propensity score methods derived through a data-driven approach and negative control experiments across ten pairwise comparisons, with results meta-analysed to generate 1280 study effects. For each study effect, we did negative control outcome experiments using a possible 123 controls identified through a data-rich algorithm. This process used a set of predefined baseline patient characteristics to provide the most accurate prediction of treatment and balance among patient cohorts across characteristics. The study is registered with the EU Post-Authorisation Studies register, EUPAS35296.
Findings
Among 1 355 349 antihypertensive users (363 785 ACEI or ARB monotherapy users, 248 915 CCB or THZ monotherapy users, 711 799 ACEI or ARB combination users, and 473 076 CCB or THZ combination users) included in analyses, no association was observed between COVID-19 diagnosis and exposure to ACEI or ARB monotherapy versus CCB or THZ monotherapy (calibrated hazard ratio [HR] 0·98, 95% CI 0·84–1·14) or combination use exposure (1·01, 0·90–1·15). ACEIs alone similarly showed no relative risk difference when compared with CCB or THZ monotherapy (HR 0·91, 95% CI 0·68–1·21; with heterogeneity of >40%) or combination use (0·95, 0·83–1·07). Directly comparing ACEIs with ARBs demonstrated a moderately lower risk with ACEIs, which was significant with combination use (HR 0·88, 95% CI 0·79–0·99) and non-significant for monotherapy (0·85, 0·69–1·05). We observed no significant difference between drug classes for risk of hospital admission with COVID-19, hospital admission with pneumonia, or hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis across all comparisons.
Interpretation
No clinically significant increased risk of COVID-19 diagnosis or hospital admission-related outcomes associated with ACEI or ARB use was observed, suggesting users should not discontinue or change their treatment to decrease their risk of COVID-19.
Funding
Wellcome Trust, UK National Institute for Health Research, US National Institutes of Health, US Department of Veterans Affairs, Janssen Research & Development, IQVIA, South Korean Ministry of Health and Welfare Republic, Australian National Health and Medical Research Council, and European Health Data and Evidence Network.
Introduction
,
,
Speculatively, angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs), both of which block the renin–angiotensin system (RAS), might affect people’s susceptibility to COVID-19 and worsen its severity. Driving this hypothesis is the mechanism by which SARS-CoV-2 enters human cells: by binding to the membrane-bound aminopeptidase angiotensin-converting enzyme 2 (ACE2), the expression of which might be altered by chronic exposure to RAS therapy.
,
,
,
,
,
,
,
,
,
Speculation about the effects of RAS therapy on susceptibility to and severity of COVID-19 has generated substantial public health concerns, resulting in the release of statements from health regulatory agencies and clinical societies advocating that, in the absence of direct evidence of harm with COVID-19, these medicines should not be discontinued.
,
However, inconsistencies in recommendations have emerged, with suggestions that users of these medicines should be monitored closely. Unlike clinical trials that are being proposed to investigate the withdrawal of ACEIs and ARBs among symptomatic patients with COVID-19, it is less likely that large-scale, population-based trials assessing susceptibility to COVID-19 among users of RAS therapy will be done in a timely manner.
,
Withholding these medicines, however, might result in worse cardiovascular outcomes, with some studies reporting an increased risk of myocardial injury resulting from illness with COVID-19.
Evidence before this study
We systematically searched PubMed, Embase, clinical trial registries, and preprint servers for research articles published from inception until March 27, 2020. No language restriction was applied. We found no investigations of the real-world safety of first-line antihypertensive medications involving COVID-19 diagnoses. Studies examining the association between renin–angiotensin system inhibitor use and COVID-19 susceptibility have since been published that report no COVID-19 risk or a lower risk associated with use of angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs). However, these studies have small sample sizes, limited confounder adjustment, or methodological limitations such as immortal time bias and collider bias. We identified one small study directly comparing the effects of ACEI versus ARB use among symptomatic patients with COVID-19 that showed no difference in patient outcomes.
Added value of this study
This study comprehensively evaluates the safety of ACEIs and ARBs in COVID-19 by examining a large number of different comparisons using state-of-the-art methods to control for residual confounding and bias across a distributed network. Our study shows similar results across three databases from two countries. ACEI and ARB use does not confer increased risk of: COVID-19 diagnosis; hospital admission with COVID-19; hospital admission with pneumonia; or hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis compared with people taking calcium channel blockers and thiazide or thiazide-like diuretics.
Implications of all the available evidence
Use of ACEIs and ARBs does not affect COVID-19 susceptibility risk, and these results are in keeping with medicines regulatory and clinical society recommendations that patients should not alter their treatment with these medicines to reduce their COVID-19 risk.
,
,
,
For example, comparing the risk of COVID-19 among users of ACEIs or ARBs with an unexposed control population can result in recruitment of non-comparable participants, confounding by indication, and the absence of a clear index date for when follow-up should start, all of which can induce bias. Reliable evidence should also be replicable, generalisable, and robust. To draw strong conclusions from observational studies, it is essential that consistent findings are produced from transparent, well designed analyses across multiple populations and data capture processes to ensure that any associations are not due to systematic error or applicable only in narrow contexts. We aimed to determine whether exposure to ACEIs or ARBs is associated with an increased susceptibility to COVID-19 among patients with hypertension.
Methods
Study design
Data sources
Two particular benefits of this standardisation are that contributing centres can participate in distributed network analyses without needing to share patient-level information and that data provenance can be ensured while applying a common analytical code across all data sources in a consistent manner. The data sources included the Columbia University Irving Medical Center (New York, NY, USA) data warehouse (CUIMC), the Information Systems for Research in Primary Care (SIDIAP) database, and the US Department of Veterans Affairs OMOP (VA-OMOP) database. CUIMC EHRs contain data, including clinical diagnoses, prescriptions, laboratory tests, demographics, and COVID-19 tests and diagnosis, from approximately 6 million cumulative patients from the New York-Presbyterian Hospital and Columbia University Irving Medical Center in the USA.
SIDIAP covers approximately 80% of the population of Catalonia (Spain), with approximately 6 million patients, and contains data collected since 2006 from general practice EHRs linked to hospital admissions, with information on diagnoses, prescriptions, laboratory tests, and lifestyle and sociodemographics, and the central database of RT-PCR COVID-19 tests.
VA-OMOP covers approximately 12 million patients from 170 medical centres across the USA and includes administrative, clinical, laboratory, and pharmacy data repositories that are linked using unique patient identifiers.
All data partners received institution review board approval or waiver in accordance with their institutional governance guidelines. Use of SIDIAP was approved by the Clinical Research Ethics Committee of the Institut Universitari d’Investigació en Atenció Primària Jordi Gol (Barcelona, Spain; project code 20/070-PCV). Use of VA-OMOP was reviewed by the US Department of Veterans Affairs Central Institutional Review Board and was determined to meet the criteria for exemption under Exemption Category 4(3) and approved the request for Waiver of HIPAA Authorization. Use of CUIMC was approved by the Columbia University Institutional Review Board as an OHDSI network study (AAAO7805).
Cohort eligibility, study period, and follow-up

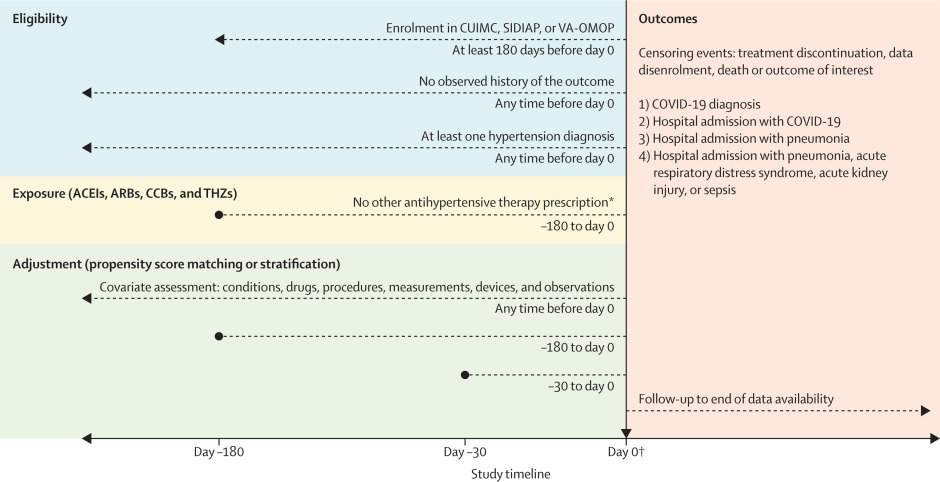
Figure 1ICARIUS susceptibility study design
We highlight eligibility criteria, exposure definitions, adjustment strategies, index date specification (day 0; horizontal black arrow), and outcome definitions and time at risk. Exposure involves prescriptions to drugs with RxNorm ingredients that map to the first-line antihypertensive drug classes: ACEIs, ARBs, CCBs, and THZs. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. ICARIUS=International Covid-19 ACE Receptor Inhibition Utilization and Safety. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership. *For monotherapy analysis only; other antihypertensive therapy included rate-limiting CCBs, diuretics, and β blockers. †Day 0 is the most recent observed prescription for target or drug comparator between Nov 1, 2019, and Jan 31, 2020.
Exposures
Outcomes
We investigated four COVID-19-related outcomes: COVID-19 diagnosis; hospital admission with COVID-19; hospital admission with pneumonia; and hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis. Positive COVID-19 PCR test results or SNOMED diagnostic codes defined COVID-19 status. COVID-19 antibody tests were not available when we did the study. The full details of the participant cohorts and outcome definitions used can be found in the protocol.
Statistical analysis
,
This process used a large set of predefined baseline patient characteristics (including age, sex, race [US data], and other demographics) and previous conditions, drug exposures, procedures, and health service use behaviours to provide the most accurate prediction of treatment and balance patient cohorts across many characteristics. For computational efficiency, we excluded all features that occurred in fewer than 0·1% of patients within the target and comparator cohorts before propensity score model fitting.
We only included estimates that passed propensity score diagnostics in the main meta-analysis, with meta-analytic estimates based on all data sources provided as a sensitivity analysis. For both monotherapy and combination use of the ACEI, ARB, CCB, THZ, ACEI or ARB, and CCB or THZ class groups (ten pairwise comparisons) to study four outcomes in three data sources (plus one meta-analysis) using two propensity score-adjustment approaches, we generated 1280 study effects.
,
For each study effect, we did negative control outcome experiments, for which the null hypothesis of no effect is believed to be true, using a possible 123 controls identified through a data-rich algorithm and validated in a previous antihypertensive comparative study.
,
Using the empirical null distributions from these experiments, we calibrated each study effect HR estimate, its 95% CI, and the p value to reject the null hypothesis of no differential effect.
We declared an HR as significantly different from no effect when its calibrated p value was less than 0·05 without correcting for multiple testing.
extensive patient characteristics to evaluate cohort balance before and after adjustment using the propensity score, negative control calibration plots to assess residual bias, and Kaplan-Meier plots to examine HR proportionality assumptions. We defined target and comparator cohorts to stand in empirical equipoise if the majority of patients in both carried preference scores between 0·3 and 0·7 and to achieve sufficient balance if all after-adjustment baseline characteristics returned an absolute standardised mean differences of less than 0·1.
Heterogeneity following meta-analysis was defined by an I2 value of more than 40%.
The prespecified ICARIUS protocol and start-to-finish open and executable source code are available online. To promote transparency and facilitate sharing and exploration of the complete result set, an interactive web application provides study diagnostics and results for all study effects.
The study is registered with the EU Post-Authorisation Studies register, EUPAS35296.
Role of the funding source
The funders of the study had no role in study design, study execution, data collection, data interpretation, writing of the report, or the decision to submit for publication, and all authors share responsibility for the decision to submit this work for publication. This was a federated data analysis, and no single author had access to all of the underlying data. TD-S, SF-B, and MA had access to the data in SIDIAP; TF and GH had access to the data in CUIMC; and SLD and KL had access to the data in VA-OMOP. DRM, MMC, SCY, GH, and MAS had full access to all aggregate results.
Results
Among 1 355 349 patients included in the analysis, 363 785 patients with hypertension who received ACEI or ARB monotherapy were compared with 248 915 patients who received CCB or THZ monotherapy, contributing 121 213 person-years and 81 261 person-years of follow-up, respectively. The overall incidence of COVID-19 diagnosis was 5·6 per 1000 person-years among patients who received ACEI or ARB monotherapy compared with 4·8 per 1000 person-years among those who received CCB or THZ monotherapy, although incidence rates varied by data source.
Corresponding patient cohort size and diagnosis incidence rates were 268 711 and 5·6 per 1000 person-years for ACEI (alone) monotherapy users and 92 485 and 5·1 per 1000 person-years for ARB (alone) monotherapy users. Cohorts for combination users (as monotherapy or combination therapy) were as large as 711 799 for ACEI or ARB users and 473 076 for CCB or THZ users.
Table 1Populations and COVID-19 diagnoses for ACEI, ARB, CCB, and THZ monotherapy and combination user cohorts
ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. MDRR=minimum detectable risk ratio. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership.
Table 2Baseline patient characteristics for prevalent use of ACEI or ARB (target) and CCB or THZ (comparator) monotherapy in the SIDIAP data source
We report the proportion of selected baseline characteristics and standardised mean difference among ACEI, ARB, CCB, and THZ users before and after propensity score stratification. Less extreme standard differences through stratification suggest improved balance between patient cohorts through propensity score adjustment. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic.
Table 3Baseline patient characteristics for prevalent use of ACEI or ARB (target) and CCB or THZ (comparator) monotherapy in the VA-OMOP data source
We report the proportion of selected baseline characteristics and standardised mean difference among ACEI, ARB, CCB, and THZ users before and after propensity score stratification. Less extreme standard differences through stratification suggest improved balance between patient cohorts through propensity score adjustment. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership.
Table 4Baseline patient characteristics for prevalent use of ACEI or ARB (target) and CCB or THZ (comparator) monotherapy in the CUIMC data source
We report the proportion of selected baseline characteristics and standardised mean difference among ACEI, ARB, CCB, and THZ users before and after propensity score stratification. Less extreme standard differences through stratification suggest improved balance between patient cohorts through propensity score adjustment. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. THZ=thiazide or thiazide-like diuretic.

Figure 2Cohort balance diagnostics comparing ACEI or ARB and CCB or THZ monotherapy prevalent use for the risk of COVID-19 diagnosis
We plotted the absolute standardised difference in population proportions of all available patient characteristics (6571 in SIDIAP, 11 183 in VA-OMOP, and 18 291 in CUIMC) before and after propensity score stratification or matching across data sources. Using stratification, CUIMC fails study diagnostics for this comparison as the absolute standardised mean difference is not consistently less than 0·1. Dashed lines indicate no before and after adjustment. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership.

Figure 3Calibrated HRs for COVID-19-related outcomes for ACEI, ARB, CCB, and THZ prevalent use across data sources
Outcomes are COVID-19 diagnosis, hospital admission with COVID-19, hospital admission with pneumonia, and hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis. We plot calibrated HRs and their 95% CIs (indicated by error bars) labelled by propensity score adjustment method. Greyed out data source entries do not pass study diagnostics and greyed out meta-analysis entries return a heterogeneity (I2) value of more than 40%. ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. HR=hazard ratio. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership.
Table 5Risk of COVID-19 diagnosis among users of ACEIs, ARBs, CCBs, and THZs
ACEI=angiotensin-converting enzyme inhibitor. ARB=angiotensin receptor blocker. CCB=calcium channel blocker. CUIMC=Columbia University Irving Medical Center data warehouse. HR=hazard ratio. SIDIAP=Information Systems for Research in Primary Care. THZ=thiazide or thiazide-like diuretic. VA-OMOP=US Department of Veterans Affairs Observational Medical Outcomes Partnership.
Discussion
,
,
,
,
,
After adjustment for the higher prevalence of cardiovascular conditions in patients with COVID-19, ACEI and ARB use was not associated with an increased risk of COVID-19 diagnosis. These case-control studies included only a limited number of covariates for model adjustment. We identified only two studies that compared the risk of COVID-19 susceptibility in ACEI or ARB users with an active comparator.
,
In this context, comparing patients with similarly indicated treatments is crucial for reducing the risk of bias resulting from confounding by indication (eg, hypertension), in which the absence of treatment indicates either too mild a disease to warrant pharmacological treatment (eg, mild hypertension under control with lifestyle and diet changes), the presence of contraindications, or extreme frailty precluding the use of preventive medicines (eg, at the end of life).
,
,
,
Indeed, de Abajo and colleagues clearly demonstrate that compared with other antihypertensive medication use, non-use was associated with a significantly reduced risk of being admitted to hospital with COVID-19, with an estimated odds ratio of 0·48 (95% CI 0·34–0·69) for severe cases and 0·57 (0·43–0·75) for less severe cases.
,
,
,
However, not all observational studies have suggested a differential effect between ACEI and ARB use.
Notably, one study comparing 124 ACEI users admitted to hospital with COVID-19 matched to 248 ARB users found no difference in the risk of 28-day all-cause mortality.
,
However, recent studies in humans have identified no difference in ACE2 levels following exposure to ACEI or ARB use.
,
,
Therefore, our findings could also be explained by residual confounding, as suggested by recent comparisons of the incidence of Staphylococcus aureus infection and other outcomes between ACEI and ARB use, which suggest that ARB use is not a perfect comparator for ACEI use, although no large-scale propensity score adjustment was used.
Although we did not observe a consistent increased risk of hospital admission with COVID-19, we did observe an increased risk of hospital admission with pneumonia, acute respiratory distress syndrome, acute kidney injury, or sepsis largely driven by ACEI use compared with CCB or THZ use. This finding might be related to the higher incidence of acute kidney injury associated with ACEI use because no increased risk was observed for pneumonia, and acute kidney injury would be considerably more frequent than acute respiratory distress syndrome or sepsis.
Our study has also been recognised by the European Medicines Agency ENCePP Guide on Methodological Standards in Pharmacoepidemiology for COVID-19 studies.
We examined outpatient prevalent use of antihypertensive therapy because a new-user design in the context of COVID-19, which has widely affected the provision of routine care, is infeasible. Therefore, mediators on the causal pathway between exposure and outcome might be included in the adjustment. However, this might not necessarily result in bias, as COVID-19 is a new illness and will not have affected the decision to initiate one drug over another. Similarly, biological mechanisms relating to ACE2 expression might require chronic exposure, hindering a new-user design. Previous treatment remains highly correlated with many baseline features that our large-scale propensity model considers when balancing patients and can provide some protection against this potential bias.
residual confounding is still possible.
Our findings stand in agreement with regulatory and clinical society advice that ACEI and ARB therapy should be continued in light of COVID-19. Furthermore, the marginal difference between ACEIs and ARBs does not warrant class switching to reduce COVID-19 susceptibility.
Contributors
DRM, MMC, SCY, and MAS conceived the study. DRM, MMC, SCY, NP, KvB, CS, MEM, CGL, FN, TMA, RWP, DP-A, PBR, GH, and MAS developed the study protocol. MMC, SCY, JW, AGS, MJS, PRR, RDW, and MAS developed analysis tools. KK, TD-S, SF-B, MA, SLD, KL, TF, and GH executed the study in their data sources. TD-S, SF-B, and MA had access to the data in SIDIAP; TF and GH had access to the data in CUIMC; and SLD and KL had access to the data in VA-OMOP. DRM, MMC, SCY, GH, and MAS had full access to all aggregate results. All authors contributed to the analysis of results and writing of the manuscript.
Declaration of interests
MAS reports grants from the US National Institutes of Health and IQVIA, and personal fees from Janssen Research & Development and Private Health Management, during the conduct of the study. DRM is supported by a Wellcome Trust Clinical Research Development Fellowship (grant 214588/Z/18/Z) and reports grants from the Scottish Chief Scientist Office, Health Data Research UK, the UK National Institute for Health Research, and Tenovus, outside the submitted work. KvB is partly funded by the European Health Data and Evidence Network project, which has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement number 806968; the Joint Undertaking receives support from the EU’s Horizon 2020 programme. SLD reports grants from Anolinx, Astellas Pharma, AstraZeneca Pharmaceuticals, Boehringer Ingelheim International, Celgene, Eli Lilly and Company, Genentech, Genomic Health, Gilead Sciences, GlaxoSmithKline, Innocrin Pharmaceuticals, Janssen Pharmaceuticals, Kantar Health, Myriad Genetic Laboratories, Novartis International, and Parexel International, through the University of Utah (Salt Lake City, UT, USA) and the Western Institute for Biomedical Research (Salt Lake City, UT, USA), outside the submitted work. GH reports grants from the US NIH National Library of Medicine, during the conduct of the study, and grants from Janssen Research, outside the submitted work. KK is an employee of IQVIA. HMK reports personal fees from UnitedHealth, IBM Watson Health, Element Science, Aetna, Facebook, Siegfried & Jensen Law Firm, Arnold & Porter Law Firm, Ben C Martin Law Firm, and the National Center for Cardiovascular Diseases (Beijing, China); ownership of Hugo Health and Refactor Health; contracts from the US Centers for Medicare & Medicaid Services; and grants from Medtronic, US Food and Drug Administration, Johnson and Johnson, and Shenzhen Center for Health Information, outside the submitted work. JCEL reports grants from the UK Medical Research Council and Versus Arthritis (UK), outside the submitted work. FN was an employee at AstraZeneca until September, 2019, outside the submitted work. AP-U reports grants from Fundacion Alfonso Martin Escudero and the UK Medical Research Council, outside the submitted work. DP-A reports grants from Amgen, UCB Biopharma, and Les Laboratoires Servier; personal fees (paid to his department) from Amgen and UCB Biopharma; and support for training programmes organised by his department from Innovative Medicines Initiative-funded European Health Data and Evidence Network and European Medical Information Framework consortia, and Synapse Management Partners. PRR reports grants from the Innovative Medicines Initiative and Janssen Research & Development, during the conduct of the study. MMC is an employee of Janssen Research & Development, during the conduct of the study. PBR, JW, and MJS are employees of Janssen Research & Development and shareholders in Johnson & Johnson, during the conduct of the study. AGS reports personal fees from Janssen Research & Development, outside the submitted work. CA, MA, TMA, CS, TD-S, TF, SF-B, CGL, KL, MEM, RWP, NP, AEW, RDW, SCY and LZ declare no competing interests.
Data sharing
Acknowledgments
Supplementary Material
References
- 1.
Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China.
JAMA Cardiol. 2020; 5: 802-810
- 2.
Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19).
JAMA Cardiol. 2020; 5: 811-818
- 3.
Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China.
Intensive Care Med. 2020; 46: 846-848
- 4.
Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19.
N Engl J Med. 2020; 382: 1653-1659
- 5.
Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus.
Nature. 2003; 426: 450-454
- 6.
SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor.
Cell. 2020; 181: 271-280.e8
- 7.
Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2.
Circulation. 2005; 111: 2605-2610
- 8.
Human intestine luminal ACE2 and amino acid transporter expression increased by ACE-inhibitors.
Amino Acids. 2015; 47: 693-705
- 9.
Localization of ACE2 in the renal vasculature: amplification by angiotensin II type 1 receptor blockade using telmisartan.
Am J Physiol Renal Physiol. 2009; 296: F398-F405
- 10.
Azilsartan ameliorates diabetic cardiomyopathy in young db/db mice through the modulation of ACE-2/ANG 1-7/Mas receptor cascade.
Biochem Pharmacol. 2017; 144: 90-99
- 11.
Cardioprotective effects of telmisartan against heart failure in rats induced by experimental autoimmune myocarditis through the modulation of angiotensin-converting enzyme-2/angiotensin 1-7/mas receptor axis.
Int J Biol Sci. 2011; 7: 1077-1092
- 12.
Upregulation of angiotensin-converting enzyme 2 after myocardial infarction by blockade of angiotensin II receptors.
Hypertension. 2004; 43: 970-976
- 13.
Telmisartan attenuates aortic hypertrophy in hypertensive rats by the modulation of ACE2 and profilin-1 expression.
Regul Pept. 2011; 166: 90-97
- 14.
EMA advises continued use of medicines for hypertension, heart or kidney disease during COVID-19 pandemic.
- 15.
Position statement of the ESC Council on hypertension on ACEI-inhibitors and angiotensin receptor blockers.
- 16.
Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection?.
Lancet Respir Med. 2020; 8: e21
- 17.
First randomised trial backs safety of common heart drugs in COVID-19 patients.
- 18.
Association of inpatient use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19.
Circ Res. 2020; 126: 1671-1681
- 19.
ACE inhibitors, ARBs and other anti-hypertensive drugs and novel COVID-19: an association study from the COVID symptom tracker app in 2,215,386 individuals.
SSRN. 2020; ()
- 20.
Response by Cohen et al to letter regarding article, “Association of inpatient use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19”.
Circ Res. 2020; 126: e140-e141
- 21.
Collider bias undermines our understanding of COVID-19 disease risk and severity.
medRxiv. 2020; ()
- 22.
Observational Health Data Sciences and Informatics (OHDSI): opportunities for observational researchers.
Stud Health Technol Inform. 2015; 216: 574-578
- 23.
Validation of a common data model for active safety surveillance research.
J Am Med Inform Assoc. 2012; 19: 54-60
- 24.
Columbia Open Health Data, clinical concept prevalence and co-occurrence from electronic health records.
Sci Data. 2018; 5180273
- 25.
Construction and validation of a scoring system for the selection of high-quality data in a Spanish population primary care database (SIDIAP).
Inform Prim Care. 2011; 19: 135-145
- 26.
Data resources in the Department of Veterans Affairs.
Diabetes Care. 2004; 27: B22-B26
- 27.
Formulae for sample size, power and minimum detectable relative risk in medical studies.
Statistician. 1992; 41: 185-196
- 28.
The central role of the propensity score in observational studies for causal effects.
Biometrika. 1983; 70: 41-55
- 29.
Evaluating large-scale propensity score performance through real-world and synthetic data experiments.
Int J Epidemiol. 2018; 47: 2005-2014
- 30.
Meta-analysis in clinical trials.
Control Clin Trials. 1986; 7: 177-188
- 31.
Interpreting observational studies: why empirical calibration is needed to correct p-values.
Stat Med. 2014; 33: 209-218
- 32.
Robust empirical calibration of p-values using observational data.
Stat Med. 2016; 35: 3883-3888
- 33.
Accuracy of an automated knowledge base for identifying drug adverse reactions.
J Biomed Inform. 2017; 66: 72-81
- 34.
Comprehensive comparative effectiveness and safety of first-line antihypertensive drug classes: a systematic, multinational, large-scale analysis.
Lancet. 2019; 394: 1816-1826
- 35.
Improving reproducibility by using high-throughput observational studies with empirical calibration.
Philos Trans A Math Phys Eng Sci. 2018; 37620170356
- 36.
A tool for assessing the feasibility of comparative effectiveness research.
Comp Eff Res. 2013; 3: 11-20
- 37.
Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research.
Commun Stat Simul Comput. 2009; 38: 1228-1234
- 38.
Therapy With agents acting on the renin–angiotensin system and risk of severe acute respiratory syndrome coronavirus 2 infection.
Clin Infect Dis. 2020; 71: 2291-2293
- 39.
Renin–angiotensin–aldosterone system blockers and the risk of Covid-19.
N Engl J Med. 2020; 382: 2431-2440
- 40.
Renin–angiotensin–aldosterone system inhibitors and risk of covid-19.
N Engl J Med. 2020; 382: 2441-2448
- 41.
Use of renin–angiotensin–aldosterone system inhibitors and risk of COVID-19 requiring admission to hospital: a case-population study.
Lancet. 2020; 395: 1705-1714
- 42.
Association of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers with the risk of hospitalization and death in hypertensive patients with coronavirus disease-19.
medRxiv. 2020; ()
- 43.
Association of use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with testing positive for coronavirus disease 2019 (COVID-19).
JAMA Cardiol. 2020; 5: 1020-1026
- 44.
Dealing with limited overlap in estimation of average treatment effects.
Biometrika. 2009; 96: 187-199
- 45.
Paradoxical relations of drug treatment with mortality in older persons.
Epidemiology. 2001; 12: 682-689
- 46.
Diagnosing and responding to violations in the positivity assumption.
Stat Methods Med Res. 2012; 21: 31-54
- 47.
Active-comparator design and new-user design in observational studies.
Nat Rev Rheumatol. 2015; 11: 437-441
- 48.
Evaluation of angiotensin-converting enzyme (ACE), its homologue ACE2 and neprilysin in angiotensin peptide metabolism.
Biochem J. 2004; 383: 45-51
- 49.
Comparative impacts of ACE (angiotensin-converting enzyme) inhibitors versus angiotensin II receptor blockers on the risk of COVID-19 mortality.
Hypertension. 2020; 76: e15-e17
- 50.
Antihypertensive medication uses and serum ACE 2 levels: ACEIs/ARBs treatment does not raise serum levels of ACE2.
medRxiv. 2020; ()
- 51.
ACEI inhibition and cardiometabolic risk factors, lung ACE2 and TMPRSS2 gene expression, and plasma ACE2 levels: a Mendelian randomization study.
medRxiv. 2020; ()
- 52.
Comparisons of Staphylococcus aureus infection and other outcomes between users of angiotensin-converting-enzyme inhibitors and angiotensin II receptor blockers: lessons for COVID-19 from a nationwide cohort study.
Wellcome Open Res. 2020; 5: 77
- 53.
Retraction: cardiovascular disease, drug therapy, and mortality in Covid-19.
N Engl J Med. 2020; 3822582
- 54.
European Network of Centres of for Pharmacoepidemiology and Pharmacovigilance. Guide on methodological standards in pharmacoepidemiology, 8th revision. Amsterdam: 2020.
Article Info
Publication History
Published: December 17, 2020
Identification
Copyright
© 2020 The Author(s). Published by Elsevier Ltd.