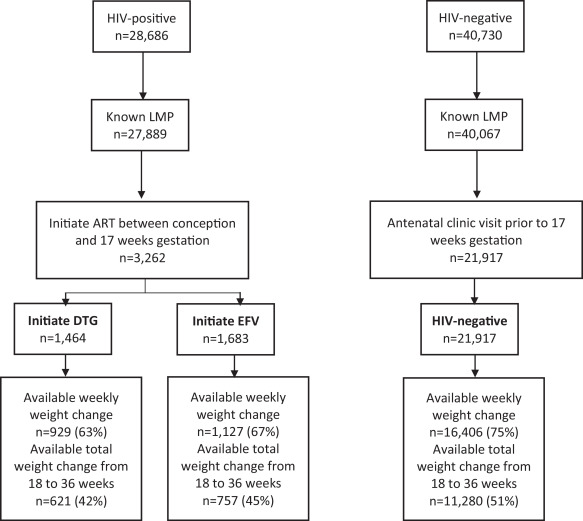
2.1 The Tsepamo study
]. Data are abstracted from the maternity obstetric record (a record of antenatal care) at the time of delivery from all women delivering at selected hospitals throughout the country. Tsepamo included 8 sites (~45% of all births in Botswana) from August 2014-July 2018 and expanded to include 18 sites (increasing coverage to ~72% of all births) from July 2018-March 2019. The maternity sites that were originally included were 2 tertiary referral hospitals, 5 district hospitals, and 1 primary-level hospital; 4 district and 6 primary-level hospitals were added in 2018. The surveillance study captures data on >99% of all births that take place at the included sites as almost all women bring their antenatal medical records (‘maternity card’) to delivery [
,
]. In Botswana, approximately 95% of women deliver at a hospital [
].
Information collected from the maternity obstetric record includes demographics, past medical history, diagnoses, hospitalizations and complications during pregnancy, medications prescribed during pregnancy, HIV history (including timing of diagnoses, ART regimens, CD4 count and viral loads), and clinical information including lab results, blood pressure, and weight measurements. All weight measurements ascertained and recorded by nurses or midwives from the time of the first antenatal care (ANC) visit to delivery are captured in the maternity obstetric record with associated dates. Self-reported pre-pregnancy weight is recorded when available. Height is measured but rarely recorded (approximately 1%) and upper arm circumference is not measured. Gestational age is documented by midwives at the time of delivery based on the estimated date of delivery (EDD). EDD is calculated at the first ANC visit using the reported last menstrual period and confirmed by ultrasound when available. If the last menstrual period date is unknown or suspected to be incorrect, fundal height measurements are used by the midwives to estimate gestational age.
Before May 2016, Botswana recommended initiation of TDF/emtricitabine(FTC)/EFV for all ART naïve adults with CD4 <350 cells/mm3 and for all pregnant women, regardless of CD4 cell count. In May 2016, TDF/FTC/DTG replaced TDF/FTC/EFV as the first-line regimen for all adults and all pregnant women and CD4 restrictions were removed. In September 2018, Botswana began to transition from TDF/FTC/DTG to TDF/lamivudine (3TC)/DTG to decrease the pill burden from 2 pills per day (TDF/FTC plus DTG) to 1 pill per day (TDF/3TC/DTG combined formulation). Women with kidney dysfunction or intolerance/resistance to TDF/FTC could access abacavir/3TC or zidovudine/3TC. TAF is not yet available in Botswana’s national HIV program.
2.4 Analysis
We examined demographic information by exposure group using sample means and proportions. For weekly weight gain and total 18-week weight gain, we fit linear regression models to estimate mean differences and 95% confidence intervals comparing women initiating DTG to women initiating EFV, and comparing HIV-uninfected women to women initiating EFV. Our models included a 3-level exposure variable (with EFV as the referent) and were adjusted for several baseline covariates: age (<25, 25–30, ≥30 years), pre-ART CD4 in pregnancy (>200 cells/μl or HIV-uninfected, ≤200 cells/μl or missing), employment (salaried, other or unknown), education (secondary education or higher, other or unknown), parity (≥1, 0 or unknown), gravidity (≥2, 1 or unknown), marital status (yes, no or unknown), site (tertiary referral hospital, other), smoking during pregnancy (yes, no or unknown), alcohol use during pregnancy (yes, no or unknown), pre-pregnancy weight (<50 kg, 50–80 kg, ≥80 kg, unknown), baseline weight in pregnancy (<50 kg, 50–80 kg, ≥80 kg, unknown), gestational age at baseline (<12 weeks, ≥12 weeks), and any medical diagnosis prior to pregnancy other than HIV (yes, no or unknown). Examples of common diagnoses prior to pregnancy include sexually transmitted infections (STI), anemia, hypertension, and asthma.
] to estimate risk ratios (an appropriate measure of association for non-rare outcomes) and 95% confidence intervals comparing women initiating DTG to women initiating EFV, and comparing HIV-uninfected women to women initiating EFV. Our models were adjusted for the same baseline covariates listed above.
We conducted subgroup analyses to evaluate effect modification by baseline weight in pregnancy (<50 kg and ≥80 kg) and by gravidity (primigravid and non-primigravid).
,
] To account for unmeasured or residual confounding by CD4 cell count, we varied how we categorized CD4 cell count in several sensitivity analyses (e.g., including a missing indicator for CD4 cell count, dichotomizing CD4 cell count as