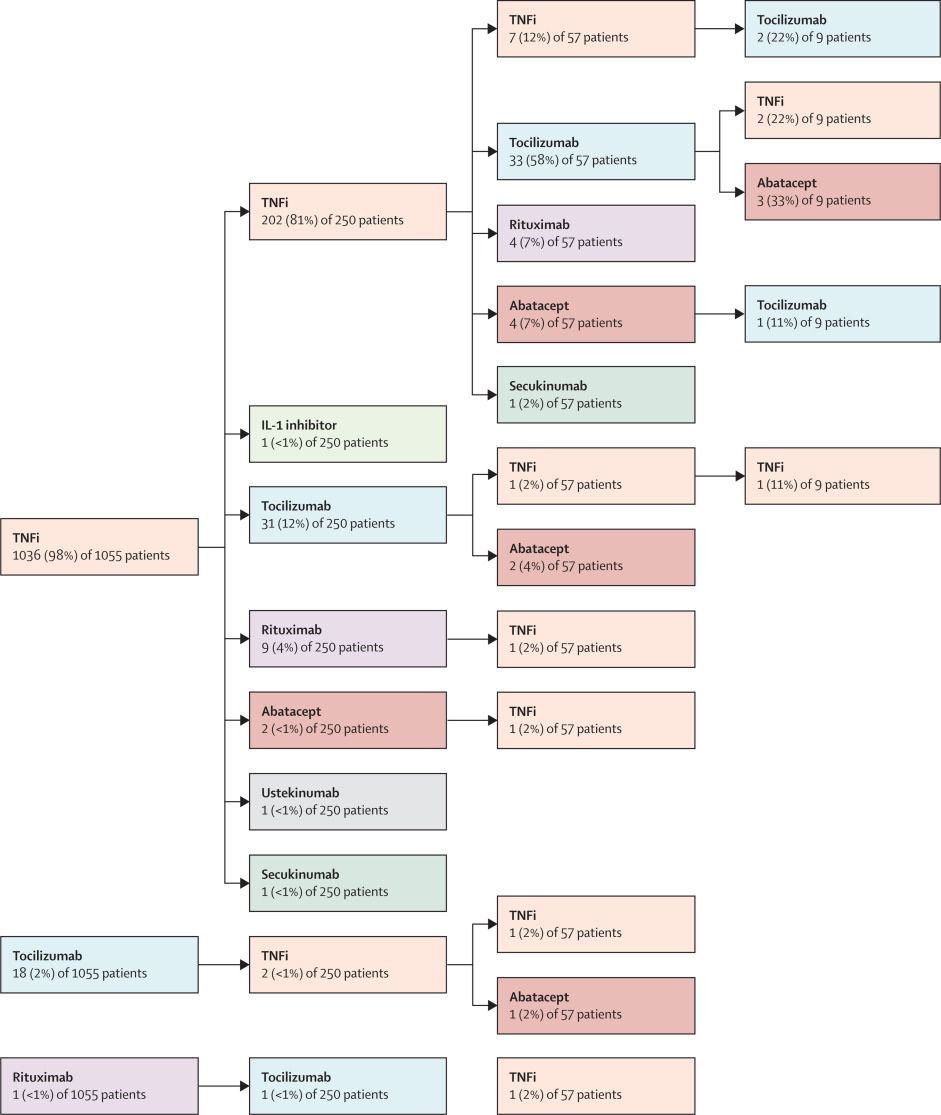
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.
‡ Propensity quintile adjusted for the following variables at the time of initiation of second biologic: second biologic start year (before 2010, 2010–2015, or 2016–2018), time since initiation of first biologic, gender, ILAR category, age, disease duration, concomitant methotrexate, concomitant steroids, active joint count, limited joint count, physician’s global assessment of overall disease activity, patient (or parent) evaluation of overall wellbeing, CHAQ, pain, erythrocyte sedimentation rate, C-reactive protein concentration, and JADAS-71 score.