(UroToday.com) Immune checkpoint inhibition has greatly improved clinical outcomes for patients with metastatic urothelial carcinoma and other cancers. Immune checkpoint inhibitors are associated with a class of adverse events, deemed immune-related adverse events due to immune activation. Nonetheless, biomarkers associated with immune-related adverse events are still lacking. Furthermore, high tumor mutational burden has been associated with better responses to immune checkpoint inhibitors in patients with metastatic urothelial carcinoma. At the 2021 GU ASCO annual meeting, Dr. Elie Akl presented the results of their study assessing the association of tumor mutational burden and immune-related adverse events. The authors hypothesized that the immune response against neoantigens is partly responsible for immune-related adverse events and investigated the association between immune-related adverse events, tumor mutational burden, and response to immune checkpoint inhibitors.
For this study, Dr. Akl identified patients with metastatic urothelial carcinoma at the Dana Farber Cancer Institute who were treated with immune checkpoint inhibitors (monotherapy or combination) and had available tumor sequencing data through Oncopanel. Tumor mutational burden was calculated using the number of non-synonymous exonic mutations per megabase. The severity of immune-related adverse events was graded using CTCAE v.5.0. A cut-off of 10/mb was assigned for tumor mutational burden. Multivariable linear regression was used to assess the relationship between tumor mutational burden, immune-related adverse events, and response.
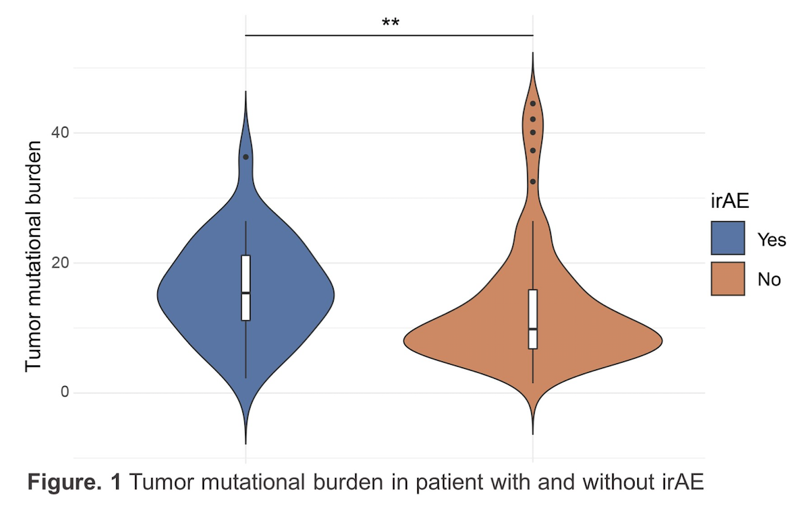
There were 101 patients with metastatic urothelial carcinoma who met the inclusion criteria, of which the majority were male (n=73), treated with single immune checkpoint inhibitor therapy (n=93), and were non-responders to therapy (n=68). There were 32 patients (32%) that reported immune-related adverse events of which 6 (6%) were grade 1 events, 20 (20%) were grade 2 events, and 6 (6%) were grade 3 events. The median time on therapy was 84 days for patients without immune-related adverse events and 88 days for patients with immune-related adverse events. Patients with immune-related adverse events had a higher median tumor mutational burden (15.4/mb) compared to patients with no immune-related adverse events (9.8/mb) (p = 0.01):

In patients on monotherapy, those with immune-related adverse events (n=27) had a higher median tumor mutational burden (15.13/mb) compared to patients with no immune-related adverse events (median tumor mutational burden 10.20/mb) (p = 0.01). Among 94 patients with radiological data, response was achieved in 16 (50%) patients with immune-related adverse events versus 10 (16%) patients with no immune-related adverse events (p< 0.001). When both immune-related adverse events and response were included in a multivariable regression, the association between immune-related adverse events and tumor mutational burden was not significant (p = 0.4). Patients with both immune-related adverse events and high tumor mutational burden had a response rate of 56% which was significantly higher than those with either immune-related adverse events but low tumor mutational burden (28.6%) or high tumor mutational burden but no immune-related adverse events (21.2 %) or low tumor mutational burden and no immune-related adverse events (10.3%) (p = 0.002).
Dr. Akl concluded this presentation with the following summary statements:
- Higher tumor mutational burden was associated with higher incidence of immune-related adverse events in patients with metastatic urothelial carcinoma on immune checkpoint inhibitor therapy
- Patients with both high tumor mutational burden and immune-related adverse events exhibited better response rates than those with only high tumor mutational burden or immune-related adverse events, suggesting that they may provide complementary tumor and host characteristics
- Further evaluation in metastatic urothelial carcinoma is needed to confirm this relationship between tumor mutational burden, immune-related adverse events, and response in a larger cohort and explore specific mutational signatures that may be associated with immune-related adverse events
Presented by: Elie W. Akl, MD, Brigham and Women’s Hospital, Boston, MA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021







![CD Projekt Red source code reportedly sells for millions in dark Web auction [Updated]](https://globalresearchsyndicate.com/wp-content/uploads/2021/02/cdpr-680x380-75x75.jpg)